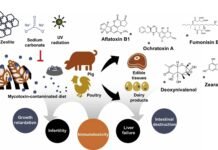
Mycotoxins, toxic secondary metabolites produced by fungi such as Aspergillus, Fusarium, and Penicillium, represent a pervasive and severe threat to global agriculture and public health. These naturally occurring toxins contaminate key agricultural commodities like maize, wheat, and barley at various stages, including pre-harvest, harvest, and storage.
Mycotoxins, toxic secondary metabolites produced by fungi such as Aspergillus, Fusarium, and Penicillium, represent a pervasive and severe threat to global agriculture and public health. These naturally occurring toxins contaminate key agricultural commodities like maize, wheat, and barley at various stages, including pre-harvest, harvest, and storage.
Regular testing of animal feed for mycotoxins is not just a regulatory requirement; it is a critical necessity to safeguard animal health, protect human consumers, and ensure the economic sustainability of the livestock industry.
Impact on Animal Health and Productivity
Aflatoxins (B1, B2, G1, G2): Primarily target the liver, causing severe hepatic damage, immunosuppression, and reduced milk and egg production. Young animals are particularly vulnerable, often exhibiting stunted growth.
Ochratoxin A (OTA): A potent nephrotoxin that leads to kidney damage and weakened immunity, especially in swine and poultry.
Fumonisins (B1, B2, B3): Disrupt sphingolipid metabolism, resulting in neurological disorders (such as ELEM in horses), pulmonary oedema in pigs, and reduced growth performance in poultry.
Zearalenone (ZEN): Acts as an oestrogen mimic, disrupting reproductive cycles and causing infertility, abortions, and stillbirths, particularly in pigs.
Trichothecenes (DON, T-2): Including Deoxynivalenol (DON or “vomitoxin”), these toxins induce vomiting, feed refusal, and gastrointestinal distress, significantly reducing productivity.
Emerging evidence highlights that the impact of mycotoxins extends beyond these effects. They can significantly alter the gut microbiome in both animals and humans, leading to dysbiosis, reduced microbial diversity, and the proliferation of opportunistic pathogens such as Escherichia coli and Clostridium species. Additionally, beneficial bacteria like Lactobacillus and Bifidobacterium are depleted, and the Firmicutes/Bacteroidetes ratio (F/ B)—a key indicator of gut health—is adversely affected.
Risks to Human Health
A major concern is the carry-over effect, where mycotoxins or their metabolites transfer from contaminated feed into animal-derived products such as milk, meat, and eggs.
For instance, Aflatoxin B1 in feed is metabolized by dairy animals into Aflatoxin M1, a Group 1 carcinogen found in milk and directly linked to liver cancer in humans. Given that milk is a dietary staple—especially for children—this risk becomes even more critical, affecting long-term public health.
Mycotoxins are often termed “silent killers” due to their chemical stability, enabling them to survive conventional food processing methods such as cooking and pasteurization.
Primary Carcinogenic Mycotoxins
Aflatoxins (B1, B2, G1, G2, M1): Strongly associated with hepatocellular carcinoma. Classified as Group 1 carcinogens by the International Agency for Research on Cancer (IARC). Aflatoxin B1 is known to metabolize into a reactive intermediate that binds to DNA, and induce mutations in the p53 tumor suppressor gene. The risk of liver cancer increases 30–60 times in individuals with concurrent Hepatitis B infection.
Fumonisins (B1, B2): Linked to oesophageal cancer and classified as Group 2B (possibly carcinogenic). They disrupt sphingolipid metabolism, leading to abnormal cell growth and apoptosis (programmed cell death) failure.
Ochratoxin A (OTA): Associated with kidney and urinary tract cancers and also classified as Group 2B. Chronic exposure is linked to Balkan Endemic Nephropathy, a kidney disease with a high incidence of associated urinary tract tumors.
Emerging Links and Secondary Risks
Zearalenone (ZEN): Associated with hormone-dependent cancers such as breast, ovarian, and cervical cancer due to its estrogenic activity.
Sterigmatocystin: A precursor to aflatoxin and a potent carcinogen affecting the liver and lungs.
Patulin: Commonly found in moldy apples; while genotoxic, its carcinogenic potential in humans is still under investigation, though experimental models suggest links to colorectal cancer.
Economic Importance of Mycotoxin Testing
The financial impact of mycotoxin contamination is substantial, with billions of dollars lost annually worldwide.
Direct losses: Reduced crop yields, animal mortality, and poor performance (lower feed conversion efficiency and weight gain).
Trade implications: Rejection of contaminated consignments, particularly in stringent markets like the EU and US.
Regulatory costs: Product recalls, legal liabilities, and long-term damage to brand reputation and consumer trust.
In the United States alone, annual losses due to mycotoxin contamination in crops such as maize and peanuts are estimated at approximately USD 932 million.
Regulatory Framework
In the regulatory landscape of animal feed and food safety, international and national bodies work together to set standards that mitigate the health and economic risks of mycotoxin contamination.
International: Codex Alimentarius Commission
The Codex Alimentarius Commission (FAO/WHO) establishes science-based global food safety standards. Codex releases the Maximum Residue Levels (MRLs): It sets recommended Maximum Levels for mycotoxins in various commodities, such as 10-15 µg/kg for aflatoxins in peanuts and 50 µg/L for patulin in apple juice. Codex relies on the Joint FAO/WHO Expert Committee on Food Additives (JECFA) to conduct scientific risk assessments that form the basis for these MLs. Its standards serve as a benchmark for international trade and are recognized by the World Trade Organization (WTO).
India: Bureau of Indian Standards (BIS)
BIS sets quality standards for animal feed and prescribes the requirements and methods of sampling for various animal feeds, such as Compounded Cattle Feed (IS 2052:2009). It has established a maximum permissible level of 20 ppb (µg/kg) for Aflatoxin in all animal feeds to prevent toxicity in livestock and carry-over into human food. BIS provides the certification mark (ISI mark) that ensures feed products comply with these safety standards before commercial sale.
India: Food Safety and Standards Authority of India (FSSAI)
FSSAI regulates food safety and has extended oversight to animal feed. It mandates compliance with BIS standards and sets strict limits for mycotoxins in food products, such as 0.5 µg/kg for Aflatoxin M1 in milk and 15 µg/kg for total aflatoxins in cereals. It publishes the Manual of Methods of Analysis of Foods (Mycotoxins), providing standardized testing procedures for regulatory laboratories across India.
The Role of Rapid Testing Methods
While reference methods like High-Performance Liquid Chromatography (HPLC) and Liquid Chromatography-Mass Spectrometry (LC-MS/MS) remain the gold standard for accuracy, they are often time-intensive, costly, and require specialized infrastructure. This has led to the widespread adoption of rapid testing methods, which are essential for on-site screening and real-time decision-making.
Key Rapid Technologies
ELISA (Enzyme-Linked Immunosorbent Assay): A cost-effective, high-throughput method delivering results within hours.
Lateral Flow Immunoassays (LFIA): Portable, easy-to-use tests providing results within minutes, ideal for on-site screening.
Strategic Advantages
Rapid testing enables real-time decision-making, allowing producers to screen raw materials at the point of entry. Contaminated batches can be immediately rejected or diverted, preventing contamination of the entire supply chain and ensuring compliance and animal safety.
Conclusion
An integrated mycotoxin management strategy—combining rapid on-site screening with precise laboratory confirmation—is essential to effectively mitigate the significant health and economic risks posed by mycotoxins. Proactive testing is not just a quality control measure; it is a fundamental pillar of sustainable and responsible livestock production.
by Dr Mrinal Kumar Sharma, Neogen Corporation